Congress seeks new guidelines on explosive fertilizer |
Urea is made when carbon dioxide is reacted with anhydrous ammonia. This process happens under intense strain, at 350 degrees urea Fahrenheit. Urea is processed to take the form of granules or stable globules generally known as prills.
How fast does urea volatilize?
Extra nitrogen is expelled from the body via urea, and since this can be very soluble, it is a very environment friendly course of. The average particular person excretes about 30 grams of urea a day, principally through urine, however a small quantity is also secreted in perspiration. It is each ammonium and nitrate which are the types of nitrogen which are most readily absorbed by vegetation. The main function of Urea fertilizer is to supply the crops with nitrogen to promote inexperienced leafy growth and make the crops look lush. Since urea fertilizer can provide solely nitrogen and never phosphorus or potassium, it’s primarily used for bloom progress.
Urea nitrate a fertilizerbased high explosive thats been used in IE devices in Afghanistan, Pakistan, Iraq, and various other terrorist acts elsewhere in the world, like the 1993 WTC bombings.Urea nitrate is produced in one step by reaction of urea with nitric acid.
— Sharanga (@Sharanga12) March 31, 2019
Dry urea may be very soluble and have to be avoided moisture until its use. Urea is unfold at rates between 40 and 300 kg/ha, but precise spreading charges will range according to farm sort and region. It is healthier to make several small to medium applications at intervals to minimize leaching losses and increase environment friendly use of the Nitrogen applied, compared with single heavy applications.
During summer, urea should be unfold just earlier than, or throughout rain to reduce attainable losses from volatilization (process wherein nitrogen is lost to the atmosphere as ammonia fuel urea n46%). Urea should not be combined with other fertilizers, as issues of physical high quality might outcome. Urea is of course produced when the liver breaks down protein or amino acids, and ammonia.
- Urea is processed to take the type of granules or strong globules generally known as prills.
- During the nitrification course of, there's an will increase the variety of free hydrogen ions (H+) in the soil, which creates acidity.
- As a result of the chemical reaction that takes place when urea is applied to the soil, special care have to be taken to make sure that the nitrogen just isn't lost when the ammonium evaporates.
Urea is crucial nitrogenous fertilizer in the market, with the best Nitrogen content material (about 46 percent). Urea is impartial in pH and can adapt to nearly all types of soils. It is a waste product fashioned naturally by metabolizing protein in people in addition to other mammals, amphibians and some fish. Urea is widely used within the agricultural sector each as a fertilizer and animal feed additive. NH2CONH2 → NH4NCO → HNCO + NH3This response is at its worst when the urea answer is heated at low strain, which happens when the answer is concentrated for prilling or granulation (see below).
Namaskar,Kindly Acknowledge the comment of Capt.Ajit #Vadakayil-
— JudgeMENT (@JUDGEisDHARMA) February 16, 2019
UREA NITRATE WAS USED IN WORLD TRADE CENTRE (NEW YORK) BOMBING IN 1993.
Urea Nitrate has a destructive power similar to ammonium nitrate explosives.
It's difficult to identify urea nitrate in post-explosion debris. pic.twitter.com/mo7xlilz1v
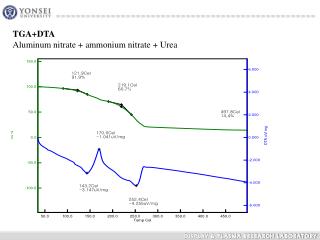
How Is Urea Made?
It passes out of the body as an answer in water, which you understand as urine. If you've a urea cycle dysfunction or a genetic disease, your physique cannot safely detoxify ammonia. As a result of the chemical reaction that takes place when urea is applied to the soil, special care should industrial urea be taken to make sure that the nitrogen is not misplaced when the ammonium evaporates. This can make urea impractical for gardeners coping with giant plots of land. The high solubility of urea also makes dry storage situations crucial.
| Комментировать | « Пред. запись — К дневнику — След. запись » | Страницы: [1] [Новые] |






